Vessel Program Curriculum Guide
Learning Objectives of the Vessel Program
Through participating in an AWRI cruise, students will:
- Experience place-based learning on the water.
- Gain exposure to the process of science through hands-on testing, observation, and interpretation of data.
- Experience a sense of belonging in the field of freshwater science.
- Understand major physical, chemical, and biological characteristics of water, and ways to scientifically analyze and observe them.
- Articulate variables and conditions that can influence water quality characteristics.
- Identify the characteristics of oligotrophic, mesotrophic, and eutrophic lakes.
- Detail how lakes differ in their characteristics, and what kind of ecosystems and human uses they can support.
- Discuss how human impacts (including invasive species) have impacted the Great Lakes and adjoining waterways.
- Name the watershed(s) that they sailed within, as well as in which they live.
- Describe the life cycle of key organisms (like midges, zebra mussels, and sea lamprey) and how they fit into the food web and larger ecosystem.

Big Ideas and Driving Questions
- How are different lakes and rivers similar and different in their physical, chemical, and biological characteristics?
- How do people use Michigan's water resources?
- How do human activities influence the quality of lakes and rivers?
- How do aquatic ecosystems play an important role in terrestrial ecosystems, and vice versa?
- How do aquatic scientists explore questions about water quality?

Michigan Science Standards
A trip on the D.J. Angus or the W.G. Jackson is an excellent way for students to have an authentic STEM experience that supports the Michigan K-12 Science Standards. Our three-dimensional learning program integrates cross-cutting concepts and disciplinary core ideas with science and engineering practices. As students do the work of scientists, they collect, analyze, and interpret water quality data. They draw comparisons about different bodies of water to ascertain patterns, explore cause and effect relationships, and develop models of systems. Educators can easily build on their students' experiences with AWRI programs through many links to their own classrooms. The Michigan-specific context of our programs stresses the unique location of our state in the Great Lakes basin, our water resources, human impacts on systems, and authentic scientific research.
The following standards are meaningfully covered on an AWRI science cruise:
4th Grade: 4-ESS2.2
5th Grade: 5-LS2-1, 5-ESS2-1MI, 5-ESS3-1
6th-8th Grade: MS-LS1-6, MS-LS2-1, MS-LS2-2, MS-LS2-3, MS-ESS2-4, MS-ESS3-3
Crosscutting Concepts: Patterns; Cause and Effect; Scale, Proportion, and Quantity; Systems and System Models; Energy and Matter: Flows, Cycles, and Conservation; Stability and Change, Interdependence of Science, Engineering, and Technology
Science and Engineering Practices: Asking Questions and Defining Problems; Planning and Carrying Out Investigations; Analyzing and Interpreting Data; Using Mathematics and Computational Thinking; Constructing Explanations and Designing Solutions
Many additional standards are touched on by our curriculum in less depth. If you need additional support in aligning our curriculum with standards for your grade level, please contact us.


Vessel Curriculum Overview
During a cruise, many activities are happening at once, and students will be absorbing a ton of information about lakes and their environment. For some students, being on a boat is itself a novel experience, let alone engaging in new and challenging science activities. Participants will maximize their experience if they have been briefed on the activities prior to the trip. Safety handouts are provided during the cruise registration process. This information is essential for the group to prepare to have a safe and successful cruise. Lessons before and after a cruise on related topics will increase students' familiarity with the topics and terminology covered on a cruise, helping them to deepen their learning and engagement with the topics.
Whether groups are sailing on the D.J. Angus or the W.G. Jackson, they will have the opportunity to be aquatic scientists. Both vessels offer the same overall trip structure and curriculum, with place-based variations appropriate to their settings. In general, cruises illustrate the biological, physical, and chemical dynamics of water, and the interrelationships of rivers, inland lakes, and the Great Lakes. The onboard curriculum is structured around conducting water quality tests and other observations to compare and contrast Lake Michigan with smaller inland lakes and rivers.
The specific topics and depth of coverage vary with age level, and AWRI Science Instructors are prepared to adapt to the needs of the visiting group. The program serves school groups as well as other organizations. It can be customized for all ages from fourth grade through adults.
Upon arrival, AWRI Science Instructors will welcome the group and orient them to the vessel and its safety features and procedures. They will also provide an introduction to where the students are, the water bodies they are visiting, and what they will have an opportunity to do onboard that day.
On a typical 2.5 hour cruise, the vessel will stop at two locations (or stations) to take water samples, conduct analysis, and make observations. Stations are sampled and analyzed on a cruise for the purpose of comparing different bodies of water. Most cruises will do one station on Lake Michigan and one on a connected inland waterway (such as Muskegon Lake or Spring Lake). If Lake Michigan is too rough, the cruise will visit other areas of the inland lakes or rivers for the second station. Shorter cruises can be provided upon request, but two stations may not be possible during shortened cruises.
Every student will have an opportunity to work directly with multiple pieces of real science equipment to make measurements and observations at each station outdoors and inside the ship's lab. AWRI Science Instructors provide students as many different experiences onboard as possible by giving new assignments at each station. For each test or observation, the instructors will orient participants to the water quality parameter being measured, the significance of this measurement to freshwater ecosystems, and the equipment and procedures to conduct the analysis. They will support the students in developing their comfort with the process of science and having success with their assigned tasks.
While the vessel is sailing between stations, science instructors share information about a range of topics, including:
- Lake food webs
- Human impacts to water quality
- Restoration and environmental clean up efforts on local and regional lakes
- Invasive species
- Research conducted by AWRI scientists
- Landmarks and place features that the vessel is sailing by
- Shoreline structures and activities, and their potential influence on water quality
- Lake stratification and temperature profiles
- Climate change and aquatic ecosystems
- Watersheds
- The significance of the Great Lakes and freshwater resources
Under the umbrella of freshwater science, particular topics can be emphasized within our curriculum upon request.
At the end of a cruise, the data collected over the course of the voyage will be analyzed and interpreted to make claims about what the data mean, and what conclusions can be drawn about the water that was sampled during the cruise. AWRI Science Instructors will facilitate review of data from each station and comparison of values from the different stations. Students will discuss actual versus expected values and trends, and practical applications of physical, chemical, and biological concepts. Instructors will answer questions from students as well as summarize key takeaways from the cruise.
Students and teachers can take their data and/or plankton and water samples (upon request) back to the classroom for further study and discussion. The data from a group's cruise can be analyzed further on its own, or in the context of other water quality data. The archive of data from AWRI cruises since 1986 is available online at https://www.gvsu.edu/wri/education/vessel-program-data-archive-59.htm. As well, the Muskegon Lake Observatory provides real-time data from April to October for comparison: https://www.gvsu.edu/wri/buoy/
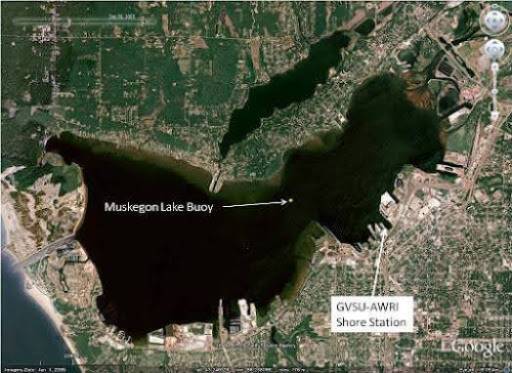
The W.G. Jackson leaves the dock at AWRI on Muskegon Lake and makes its way across the lake to Lake Michigan. Muskegon Lake is 4,149 acres or about 6.5 square miles in size. Many of the industrial areas along the shoreline of Muskegon Lake are being restored to pre-industrial conditions or re-developed. The area once had active logging and numerous industrial operations. Homes and marinas also surround the lake. Significant heavy industrial activity along with wastewater discharge, combined sewer overflows, and urban runoff in the past have led to contamination. In 1987, Muskegon Lake was designated an “Area of Concern” under the Great Lakes Water Quality Agreement. Through concerted restoration efforts since, the lake is very close to being delisted and has much higher water quality today. The Muskegon River watershed drains into Muskegon Lake and influences the water quality. Besides Muskegon and Big Rapids, the watershed has no large cities and is relatively undeveloped.
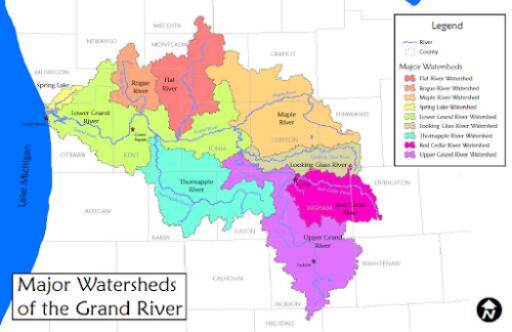
The D.J. Angus makes a trip to Lake Michigan via the Grand River. Along the route to Lake Michigan, there is a former sand mining operation, a large commercial unloading dock, marinas, and a former power plant. The headwaters of the Grand River are near the city of Jackson and the river also flows through the cities of Lansing and Grand Rapids. Besides urban areas, land use in this watershed is largely devoted to agriculture with relatively few natural areas. The other sampling point for the D.J. Angus is usually in Spring Lake, which is connected to the Grand River. Spring Lake is surrounded mainly by homes and has experienced elevated nutrient levels.
Lake Michigan has a surface area of about 22,300 square miles making it the third largest Great Lake. The flushing time of the Lake (or the average amount of time that water spends in the lake) is 62 years. The average depth of Lake Michigan is 279 feet with a maximum depth of 923 feet, making it the second deepest Great Lake. In contrast, Spring Lake's surface area is about 2 square miles with inputs of water mainly through springs, streams, and precipitation. Spring Lake is connected to the Grand River. Muskegon Lake is about 6.5 square miles in size, and it receives flow from the Muskegon River as well as tributaries such as Ryerson and Ruddiman Creeks.
Muskegon Lake
Spring Lake
What is Water Quality?
Water quality is defined in terms of physical, chemical, and biological parameters with respect to a certain use. For instance, acceptable water quality for warm water fishes would not be optimal for cold water fishes, and standards for drinking water differ from those for boating and recreation. No single factor alone indicates good water quality, and water quality in a body of water can vary with the season and location. Long-term water quality measurements from well-defined locations are needed to tell if conditions are changing or remaining the same. Many of the procedures performed on the vessel help us to understand the quality of the waters being sampled.


One way of comparing lakes and evaluating water quality is by considering the trophic status or biological productivity. Eutrophication, or aging of lakes, progresses through various trophic states (oligotrophic → mesotrophic → eutrophic). Nutrient levels, organic matter content, dissolved oxygen levels, and water transparency give clues to the trophic state or biological productivity of a water body. A trophic scale has been specially designed for use on the AWRI vessels. By evaluating data from various parameters, sampling locations are rated as O (oligotrophic), M (mesotrophic), or E (eutrophic).
Oligotrophic lakes are characterized by low nutrient levels, low biomass, high oxygen concentrations, and high transparency. Oligotrophic lakes are usually deep. Eutrophic lakes are highly productive with high nutrient levels, high biomass, low oxygen concentration in the bottom waters, and low transparency. The large volume of organic matter accumulated in bottom sediments depletes oxygen as it decomposes. Mesotrophic lakes are between the other two trophic states in their characteristics. The open waters of Lake Michigan are oligotrophic and some nearshore areas are mesotrophic.
The vessels use Van Dorn water sampling bottles to collect samples at various depths. These special bottles consist of an open-ended clear plastic cylinder that can be attached to a steel cord and lowered to any desired depth. A deckhand operates this equipment, but when conditions permit, will select 1-2 students to assist with this task. The bottles also contain thermometers to record the temperature of the water at the location of each Van Dorn bottle. Water samples from each bottle are taken into the ship's lab for analysis. On a typical cruise, two Van Dorn bottles are used: one 1 meter below the water surface and one 1 meter above the lake bottom. This allows us to compare and contrast the characteristics of the top and bottom of the water column. On more advanced or longer cruises, a third bottle may be added to sample at half the station depth.
Summary of Water Quality Parameters
With every water quality parameter, participants will explore 1) what the parameter is, 2) why it is important for us to gather information on, 3) factors which influence it, and 4) how we are measuring it. Contextual parameters that could influence our readings are also recorded, such as percent cloud cover, wave height, water depth, wind speed, wind direction, and air temperature.
Chemical Characteristics of Water Quality
Chemical characteristics of water quality measured on the vessel include: pH, conductivity, dissolved oxygen, alkalinity, and nutrients.

The pH scale is a series of numbers ranging from 0 to 14 which denote various degrees of acidity. A natural body of water can be acidic, neutral, or basic. Many factors influence pH, including the composition of the material forming the basin holding the water, acidity of rain falling into the water, and the condition of water flowing into the body of water from streams, rivers, or storm runoff. A pH between 6.7 and 8.6 will support a well-balanced fish population, though different species prefer different pH values within this range. Very few species can tolerate pH values less than 5.0 or greater than 9.0. Lake Michigan water samples typically have a pH range of 7.0 to 8.6. A pH meter is used to measure pH on the vessels.
Conductivity, or specific conductance, is the measure of the water's ability to conduct an electrical current. Conductivity depends upon the number of ions, or charged particles, in the water. Factors that influence conductivity are human activities such as road salt, agriculture, and septic systems. Conductivity influences what organisms can live in the water and can be an indicator of other kinds of pollution. A conductivity meter is used to measure this parameter.
Dissolved oxygen measures how much oxygen is available to organisms in the water. Most aquatic organisms are highly dependent upon dissolved oxygen and will experience stress when levels fall below about 3ppm (parts per million). Some fish, such as salmon and lake trout, have high oxygen needs while bullhead and carp can tolerate very low oxygen conditions.
The distribution of dissolved oxygen within an aquatic environment varies horizontally, vertically, and with time. Its distribution is dependent upon atmospheric contact, wave and current actions, thermal phenomena, waste inputs, biological activity, and other characteristics of a lake or stream. Dissolved oxygen levels are temperature and pressure dependent; cold water has the capacity to hold more oxygen than warm water. Photosynthesis by plants contributes to an increase in dissolved oxygen levels during the day. However, there are biological processes in water that consume oxygen such as respiration by organisms and decomposition of organic matter by microorganisms. In summer, there are times when the dissolved oxygen at the bottom of Spring Lake approaches zero. During spring and fall turnovers, horizontal and vertical variation of dissolved oxygen is likely to be less than during the summer.
Onboard, we use a chemical titration, the Azide Modification of the Winkler Method, to measure dissolved oxygen. Personal protective equipment (goggles and gloves) must be worn during this test.
Alkalinity is a measure of the capacity of water to neutralize acids. This is also known as the buffering capacity of water, or the ability of water to resist a decrease in pH when acid is added. Acid additions generally come from rain or snow, though soil sources are also important in some places. Alkalinity increases as water dissolves rocks containing calcium carbonate, such as calcite and limestone. Algae and industrial activity can also influence alkalinity. When a lake or stream has low alkalinity, a large influx of acids from an intense rainfall or rapid snowmelt event could result in a drop of the pH of the water, which could be harmful for organisms or human uses.
Michigan experiences acid rain (precipitation with a pH less than 5.6), yet lakes in our area are not very negatively impacted - why? The limestone in the Lake Michigan basin is a natural buffer that increases the alkalinity of waters in our area and helps to maintain soil and water pH near or above neutral. By contrast, Lake Superior has more granitic bedrock, which results in lower alkalinity in the basin’s lakes, and greater susceptibility to negative impacts from acid rain.
Nitrogen and phosphorus are nutrients that are natural parts of aquatic ecosystems. They are necessary for the growth of algae and aquatic plants, which provide food and habitat for fish, shellfish, and smaller organisms that live in water. But elevated levels of nutrients can lead to eutrophication, algae blooms, and low oxygen conditions. Nutrients in lakes can be increased by human activities such as agriculture, urban runoff, and sewer discharge. Nutrients are measured on advanced cruises only, but their impacts and implications are discussed on all.
Physical Characteristics of Water
Physical characteristics of water quality measured on the vessel include: water clarity, water color, water depth, and water temperature.

How far can we lower the disk and still see the black and white pattern? Secchi Disks are used by scientists all over the world to measure water clarity. We see readings of 5 meters or more on Lake Michigan, but as low as .5 meters in Spring Lake. Readings also vary within the same lake depending on the time of year. For additional ways to measure water clarity, we also use a turbidity tube and a turbidity meter. Clarity, or transparency, tells us about how deep light can penetrate into the water, which determines how much photosynthesis can happen in a lake. Water clarity is related to amounts of suspended particles (turbidity) as well as phytoplankton and zooplankton.
The Forel-Ule Color Scale is a standardized method of determining color of a body of water. On our vessel, we demonstrate how to use the scale, and the students get hands-on experience using it to determine the water's hue. Muskegon and Spring Lake tend to be greener, while Lake Michigan is often a brilliant blue. Water color is influenced by organic and inorganic material and life in the water, as well as sun angle, water vapor, and clouds at the time of the reading.
Many water quality parameters, such as temperature and dissolved oxygen, vary with depth as well as with the time of day. The depth of light penetration, which is influenced by turbidity, has an effect on the productivity of plants and algae in an aquatic ecosystem. Various depths in a lake or river host different assemblages of benthic (bottom-dwelling) organisms. Plankton and fish move from one depth to another based on changing environmental conditions. The boats have meters that determine the water depth, and the reading will be announced by the Captain once the vessel is officially on station.
One of the first things you might want to know about a lake when deciding to swim in it is the water temperature! This characteristic is extremely important for water quality as well. Temperature alone impacts organisms in the water, but temperature also influences other water quality parameters. Temperature of top and bottom samples is read in degrees Celsius (°C) from the Van Dorn water bottle samples.
Another device for collecting temperature data is a "fish finder" data logger that is attached to the line on the PONAR sampler. This device takes a reading every 5 feet of water depth to allow a more detailed picture of how temperature changes with depth, called a temperature profile. Temperature profiles help scientists understand temperature patterns at various depths and the relationship to the seasons of the year.
Biological Characteristics of Water
Biological characteristics of water quality measured on the vessel include: sediment and benthic organisms and plankton.

On AWRI’s vessels, the PONAR grab sampler helps teachers and students explore the mysteries of West Michigan aquatic ecosystems firsthand, by taking a bite out of the sediment at the bottom of the lake and bringing it up for us to view. We can tell a lot about a lake by the kind of material the bottom is made of, and who is living in or on it. Muskegon and Spring Lake tend to have dark, mucky sediment, while Lake Michigan is sandy. Poking through the sediment looking for living organisms is the highlight of a cruise for many passengers. We often find bloodworms (the larval form of the midge fly), phantom midges, tubifex worms, biting midges, and zebra and quagga mussel shells (or infrequently, living mussels).
Plankton are microscopic organisms distributed throughout the lake. They are found at all depths and have both plant (phytoplankton) and animal (zooplankton) forms. Both the density and the diversity of plankton are important to scientists. Plankton density is a measure of how much plankton is in the water, while diversity refers to how many different species can be observed. Plankton show a distribution pattern that can be associated with the time of day and seasons.
Plankton is the base of the food chain for Lake Michigan. The phytoplankton are the producers, and include green algae, cyanobacteria, and diatoms. Cyanobacteria, such as Microcystis, prefer warm water and high nutrients. Some cyanobacteria can fix nitrogen and produce toxins, which can present issues during a harmful algal bloom (or HAB). Zooplankton are the consumers and include tiny crustaceans such as water fleas (Daphnia), cyclops, and copepods.
A plankton sample will be gathered using a net with very fine mesh that allows water to pass through but collects the plankton organisms. This concentrated sample will be measured for density using a graduated cylinder with a Secchi disk pattern at the bottom, and later viewed under a microscope to see what species can be identified. This is often among the most exciting parts of a cruise, to witness the profusion of life found in a small drop of water!
Lake Stratification
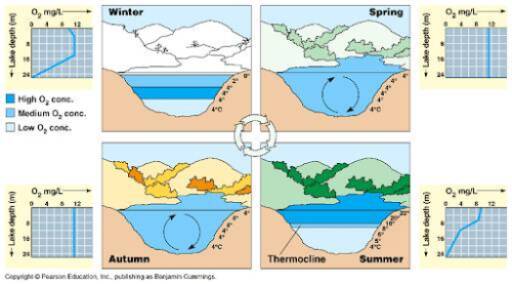
Temperature profiles sometimes show that the warm water surface layer often ends abruptly, with the temperature going rapidly from warm to cool, which is called a thermocline. The thermocline is the zone where there is a rapid temperature change, and thermoclines are observed when a lake is stratified. Lake stratification refers to the formation of distinct layers of water that do not mix with each other due to density differences. In temperate climates, many lakes are stratified during the summer, with large temperature differences between the layers of water.
As heat from the sun increases during the summer, the upper waters of the lake become warmer and lighter than deeper waters. One very important consequence of summer stratification is that circulation due to wind action is largely confined to the upper layer of water. Because the lower water mass is isolated from the atmosphere and receives little, if any, sunlight, dissolved oxygen is not replenished in this water mass. The dissolved oxygen may diminish to such a level that it limits aquatic life.
By contrast, in the fall and spring, the temperature of the lake is essentially uniform from top to bottom. This occurs when the power of the sun is reduced such that wave action on the surface will mix oxygen in the air with the water. The oxygen-rich water is driven down to lower depths, and the bottom oxygen-poor water will be brought to the surface where it can be replenished with oxygen. Turnover is also not uniform in a lake, and strong winds can cause upwellings where nutrient rich deep water moves towards the surface.
Muskegon Lake tends to follow this pattern. Since this occurs twice per year in spring and fall, these kinds of lakes are said to be dimictic; “di” meaning two and “mictic” meaning “to mix”. Because Spring Lake is shallow and a riverine system, the stratification breaks down several times throughout the summer. Every time this happens, the lake turns over. Since it mixes many times per year, we call it polymictic. Since Lake Michigan never completely freezes over, it technically only mixes once per year (fall), making it monomictic.
Climate Change
The Great Lakes and the region are influenced by larger climate change patterns affecting North America and the world. Global climate change caused by greenhouse gas emissions (such as carbon dioxide and methane) is well established by scientific study. Scientists expect that these atmospheric changes will result in increases in global air and water temperatures, extreme weather events and increased climate variability, and biotic changes. In the West Michigan region, increased variability in precipitation and changes in frequency and intensity of storms and floods are anticipated. A wetter climate is predicted, except during the summer where drier conditions are anticipated. We have already begun to observe some of these changes.
These global and regional changes influence lakes in many ways. Freshwater aquatic ecosystems are complex assemblages of species that have evolved over many thousands of years, and are particularly vulnerable to rapid changes due to climate change. Many of the parameters that we observe and test for on AWRI cruises will be impacted in different ways by climate change. Climate change also causes decreased predictability and increased uncertainty in how systems will respond to change.
Many of the most severe impacts of climate change can be mitigated if our society takes action now to reduce carbon emissions. We can also reduce the vulnerability of aquatic ecosystems to climate change by minimizing other pressures, such as reducing habitat destruction and restoring habitats, preventing the spread of invasive species, and sound stormwater management.
There are a dozen GVSU faculty members with labs at AWRI, who are doing research on a wide variety of topics within the field of aquatic science:
- Stuart Jones, Ph.D., AWRI Executive Director - Aquatic Ecology
- Bopaiah Biddanda, Ph.D. - Microbial Ecology
- Amanda Buday, Ph.D. - Conservation Social Science
- Sarah Hamsher, Ph.D. - Phycology
- Mark Luttenton, Ph.D. - Aquatic Ecology
- Jim McNair, Ph.D. - Ecological Modeling
- Ryan Otter, Ph.D. - Restoration, Pathogens, and Food Web Contaminants
- Charlyn Partridge, Ph.D. - Molecular Ecology
- Carl Ruetz, Ph.D. - Fisheries Ecology
- Alan Steinman, Ph.D. - Aquatic Ecology, Ecosystem Restoration
- Kevin Strychar, Ph.D. - Climatology
- Sean Woznicki, Ph.D. - Watershed Hydrology and Geospatial Analytics


