Christopher Lawrence

Professor of Chemistry
Office: 327D Padnos Hall
Telephone: (616) 331-2322
E-Mail: [email protected]
Education/Professional Training
B.S. 1998, University of California-Los Angeles
Ph.D. 2003, University of Wisconsin-Madison
Research Interests:
My research has focused on using vibrational spectroscopy to probe the dynamics of molecules. The environment that a molecule is in can have a great influence on its vibrational frequency. For example, when a water molecule is in the gas phase, it has a vibrational frequency of 3700 cm-1. However, in the liquid, the frequency shifts to an average of 3400 cm-1. This effect in this case stems from the very strong hydrogen bond formed with neighboring molecules. The absorption line also becomes quite broad in the liquid. This is due to the fact that not every molecule interacts in the same way with its neighbors. Those that have stronger interactions will have a greater shift in frequency than those that happen to have weaker interactions.

This figure shows the three vibrations of HOD in liquid water (the bend at the lowest frequency, the OD stretch in the middle, and the OH stretch at the highest frequency). The vertical lines indicate the value in the gas phase and the solid lines show what is observed in the liquid phase. The dashed lines are the results of a computer model that I developed.
My interest in this comes from the fact that this frequency will change in time as the molecules move around. That is, a molecule that has a strong hydrogen bond now may have a weak one some time later. Thus, by observing how these frequencies change in time, we can learn quite a bit about the dynamics of the molecules, such as the rate at which hydrogen bonds break and reform in liquid water.
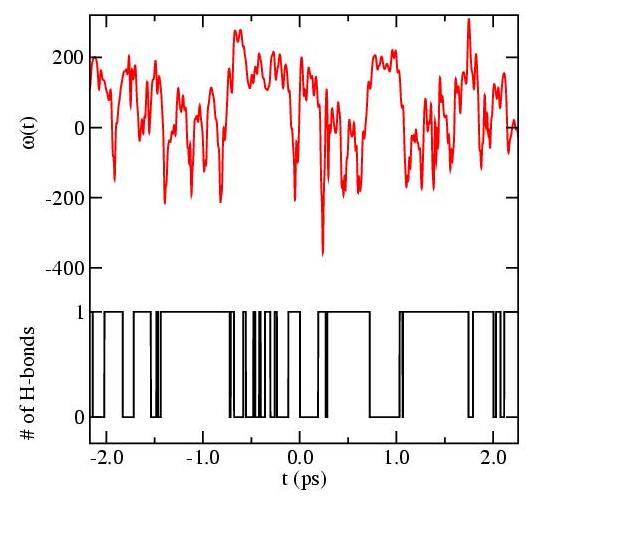
This figure shows the shift of the OH bond frequency from its average value in HOD as a function of time. Also shown is the number of hydrogen bonds to the H atom.
The drawback is that the experiments that probe this time-dependent frequency are often difficult to interpret. Thus, I assist in this interpretation by using molecular dynamics simulations to model the experiments.
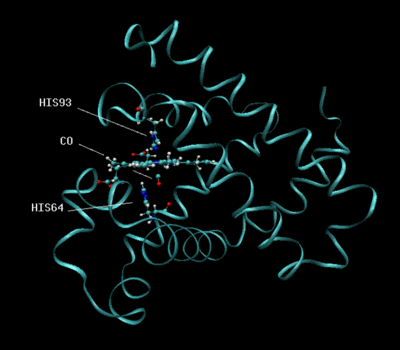
Most recently, I have been doing such modeling on carbonmonoxy myoglobin. The rationale behind choosing this system is that the vibrational frequency of the carbon monoxide is substantially different from the rest of the protein. Thus, in an experiment, one can measure this frequency as it fluctuates in time without having to worry about interference from other parts of the molecule. From this data, we hope to learn more about the dynamics of proteins.
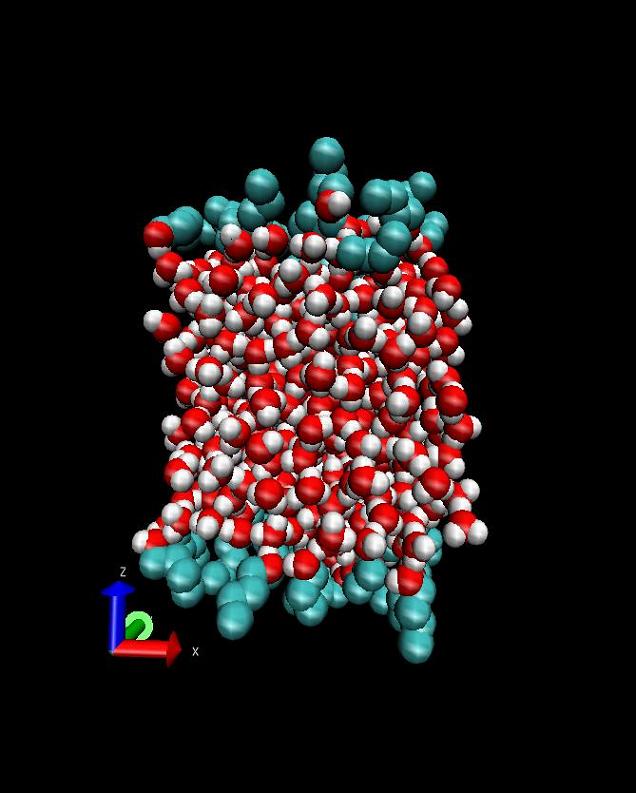
I have also recently become interested in modeling gas-liquid interfaces. Experiments have shown that when large organic molecules are added to the surface of liquid water, the rate of water evaporation drops tremendously (by a factor of 10,000). However, if the organic molecules are smaller (chains of only about four carbons), there is no change at all. To try and understand this, we are exploring the mechanism by which water evaporates through organic molecules using molecular simulations.
Butanol/water interface
Select Publications:
- Ultrafast infrared spectroscopy probes hydrogen-bonding dynamics in liquid water, C. P. Lawrence and J. L. Skinner, Chem. Phys. Lett. 369, 472 (2003).
- Quantum dynamics of simple fluids, C. P. Lawrence and J. L. Skinner, J. Chem. Phys. 120, 6621 (2004).
- On the mode-coupling theory of vibrational line broadening in near-critical fluids, C. P. Lawrence and J. L. Skinner, J. Chem. Phys. 120, 8651 (2004).
- Spectral diffusion in a fluctuating charge model of water, S. A. Corcelli, C. P. Lawrence, J. B. Asbury, T. Steinel, M. D. Fayer, and J. L. Skinner, J. Chem. Phys. 121, 8897 (2004).
